The First Fuel of Life: How Did ATP Production Evolve?
Our cells, the fundamental units of life, derive their energy from a molecule called ATP (Adenosine Triphosphate). But billions of years ago, on a "prebiotic" Earth, the sophisticated enzymes we see today didn't exist. So, how did the earliest forms of life power themselves?
A recent study published on bioRxiv sheds light on a critical "missing piece" in the origin of life: how ancestral proteins managed to produce energy with a very limited toolkit.
Starting Life with a Restricted Alphabet
Today’s proteins are built from a repertoire of 20 different amino acids. However, on prebiotic Earth, this diversity was significantly lower. Researchers reconstructed an ancestral enzyme variant named Arc1-12KR, using a restricted alphabet of only 10 prebiotic amino acids plus lysine and arginine.
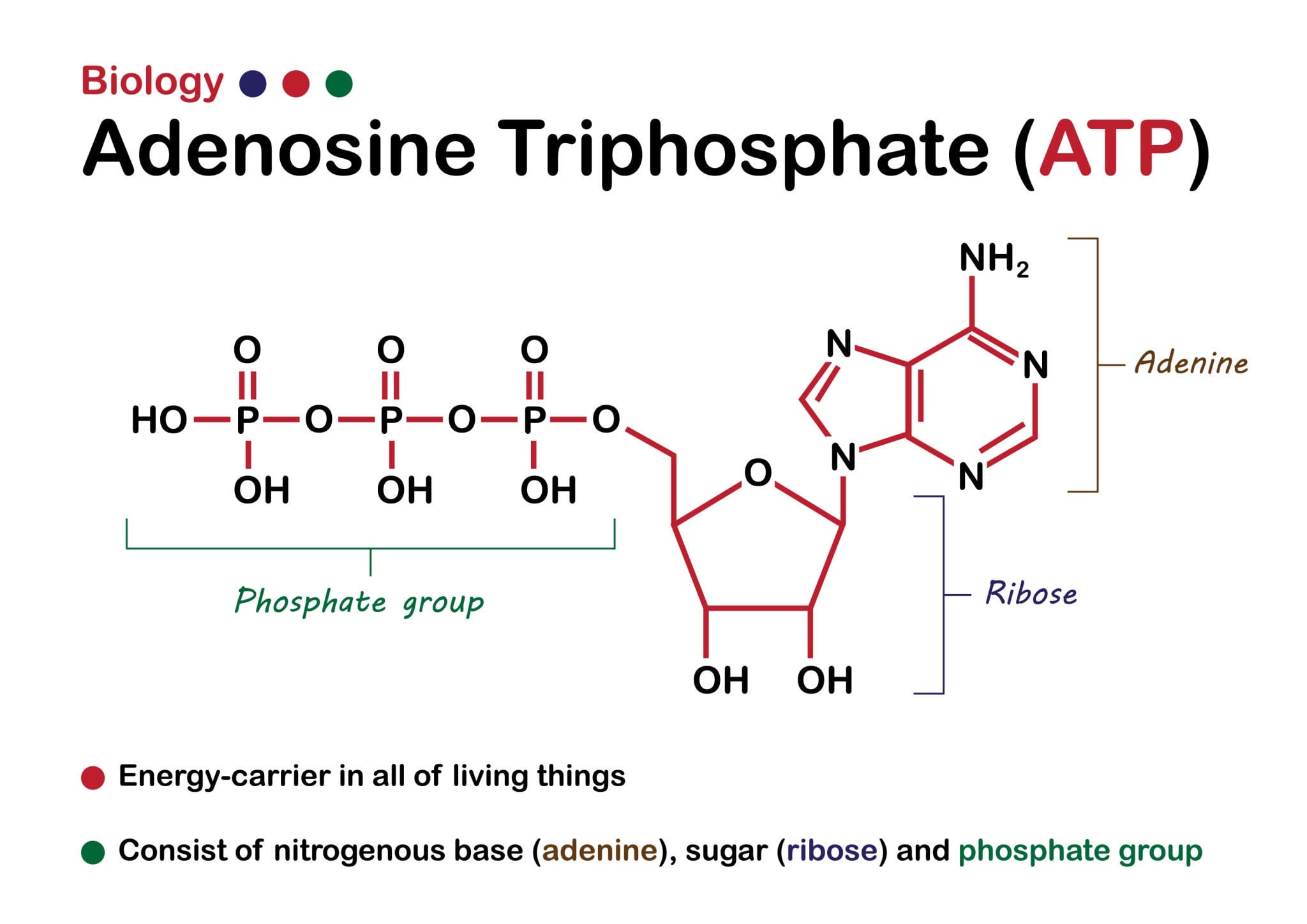
This experiment proves that life could achieve complex catalytic feats even with a "limited alphabet."
A Different Path: 2ADP → ATP + AMP
In modern organisms, enzymes like Nucleoside Diphosphate Kinases (NDKs) follow highly sophisticated pathways to synthesize ATP. However, this "ancient" enzyme variant uses a different mechanism: Disproportionation.
Essentially, it takes two "low-energy" ADP molecules and reshuffles them to create one "high-energy" ATP and one AMP. While this mechanism differs from canonical NDKs, it is chemically identical to the reaction catalyzed by modern Adenylate Kinases—suggesting a fascinating convergence on a primordial energy-generating chemistry.
The Dance of Arginine and Magnesium
Using X-ray crystallography and molecular dynamics, the study revealed that the active site of this ancient enzyme has a unique architecture. Aspartate and arginine residues cooperatively coordinate Magnesium ($Mg^{2+}$) ions.
Even under extreme constraints, this structural organization allows the enzyme to remodel its active site and facilitate noncanonical phosphoryl-transfer, effectively creating ATP where modern logic says it shouldn't.
The Evolutionary Trajectory: From Simple to Complex
The research outlines a clear evolutionary path for energy production:
- Prebiotic Era: Kinase-like proteins working with a limited amino acid repertoire, generating ATP through ADP disproportionation.
- LUCA Era (Last Universal Common Ancestor): As the amino acid alphabet expanded, modern NDKs and Adenylate Kinases emerged.
- Modern Era: The evolution of rotary ATP Synthases, the massive molecular motors that enable highly efficient ATP production from ADP and inorganic phosphate.
Conclusion
These findings demonstrate that restricted amino acid availability could actually drive the remodeling of active sites, promoting unique chemical reactions that paved the way for life. Even when arginine was replaced by lysine, the system retained its activity—showing that life, from its very inception, was built to be resilient and resourceful.
Source: biorxiv.org (Evolutionary trajectory of ATP-generating enzymes)